Corrosion and electrolysis on pleasure boats

The presence of direct and alternating current, regular electrical connections to quayside terminals, and the fact that boats spend their time immersed in water (which can be assimilated to a conductive solution), are all factors at the origin of corrosion and electrolysis phenomena.
Although these phenomena are similar, their origins are very different.
Well-known to yachters, these phenomena can have disastrous consequences. This applies to all aluminum boats made of carbon composite, wood, or GRP.
The principle behind galvanic corrosion
Galvanic corrosion requires no external power source to trigger. It has been known since the 18th century (Luigi Galvani), long before electricity storage and production systems were invented.
Three conditions must be met for this to occur. It would be best to have metals (or alloys) of different natures, an electrical connection, and a liquid to act as electrolytes. All these conditions are met in a boat. They're made up of materials and metals of different natures, they're linked together by conducting wires, and seawater acts as the electrolyte.
Once the bond is established, matter is transported from the less noble metal to the more noble. This transport takes the form of metal ion transfer.
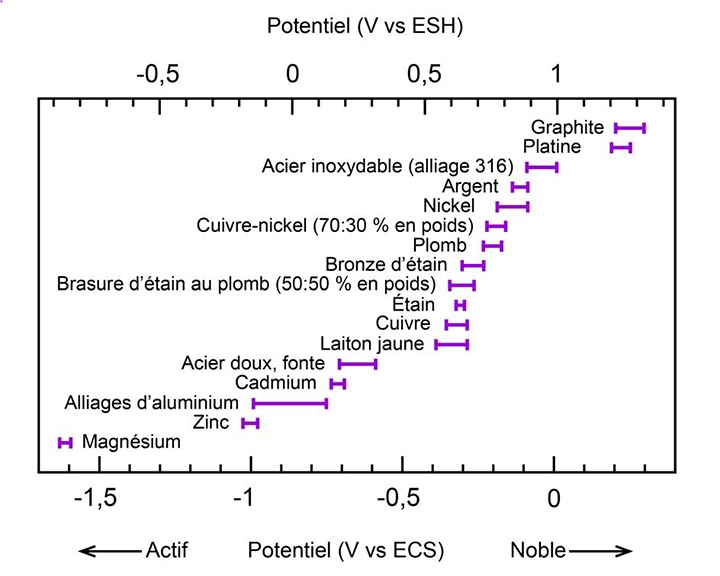
A galvanic potential characterizes each alloy or metal. Titanium, graphite, and 316 stainless steel are at the top of the list, while zinc (the metal used in anodes) is at the bottom. The greater the potential difference between the metals in contact, the greater the risk of corrosion.
This happens when a zinc anode (with a low galvanic potential) corrodes and protects an aluminum baseplate or stainless-steel propeller shaft. Hence the term sacrificial anode.
On the boat, this phenomenon can be found on the hull and inside, in the presence of stagnant water in the bilges.
Carbon composite hulls are also at risk, as their galvanic potential is very high.
Hull passes on wooden hulls and paints on steel and aluminum hulls suffer when their sacrificial protections are too large or oversized. The wood's lignin disintegrates, and the paint blisters.
On a larger scale, such as that of a port, all submerged boats may react like the elements of a battery. The presence of current further accentuates the effects of this phenomenon. And there are current: 230 volts from the quayside, 12 or 24 volts from the batteries, or 230 volts produced on the boats by the generators.
Galvanic isolators - Effective protection
Finally, when boats are connected to the 230 volts of the quay, they share a common ground through the quay line. This connection can damage neighboring vessels by destroying the less noble submerged metal parts, such as aluminum bases.
The AC circuit (230 Volts) is considered separate from the DC circuit (12/25/48 V). In reality, standards require the shore, sea, and boat earth...
Conditions are ripe for galvanic corrosion to occur.
To separate these two circuits, we recommend installing a galvanic isolator as close as possible to the shore power inlet in the boat. This is an inexpensive and easy-to-install ground line isolator, which will reduce stray currents.
More sophisticated, the isolation transformer ensures galvanic isolation between the quay's AC circuits (230 Volts) and the boat. It is indispensable on-board aluminum ships, one of the least noble metals in galvanic insulation.
Another effective protection against leakage currents is staying in the boat as it is connected to the quay. This is only sometimes possible.
Leakage currents - Origin and protection
Apart from the problem of shore power, all pleasure boats are equipped with DC and many AC networks.
These networks often suffer from leakage currents, which flow in unexpected directions.
On a DC system, the leakage current path is simple: all grounds are connected by a grounding braid to a plate in contact with seawater. The ground braid can also be brought into contact with the water via the motor and propeller shaft. In these cases, galvanic corrosion is compounded by DC electrolytic corrosion, which considerably accentuates its corrosive effects.
To avoid this, it is necessary to use bipolar electrical networks (networks that do not use metal grounds as conductors). Particular attention must be paid to diesel engines, whose starters often use the engine's mass as a conductor, allowing leakage to the sea via the propeller shaft.
Some flexible couplings, such as the Aquadrive, interpose a thick elastic washer that acts as a vibration damper and, in this case, as an insulator.

It is, therefore, advisable to be able to measure these leakage currents regularly to detect any abnormal drift.
Aware of this issue, Dolphin Charger has been offering a panel-mounted instrument for over 20 years: the Leakage Monitor 12/24 Vdc, which measures leakage currents between the battery's + terminal and the hull or directly between the - terminal and the hull. Green LEDs indicate low leakage (less than 4mA), orange, medium leakage (between 4mA and 8mA), and red, high leakage (over 8mA). Leakage currents over 10mA are often considered critical.
Once you've been alerted to a current leak, you can start investigating the cause.



